
Water | Free Full-Text | Aluminum-Doped Cobalt Ferrite as an Efficient Photocatalyst for the Abatement of Methylene Blue

Roadmap for Sustainable Mixed Ionic‐Electronic Conducting Membranes - Chen - 2022 - Advanced Functional Materials - Wiley Online Library

A layered g-C3N4 support Single-Atom Fe-N4 catalyst derived from hemin to Activate PMS for Selective degradation of electron-rich compounds via singlet oxygen species - ScienceDirect
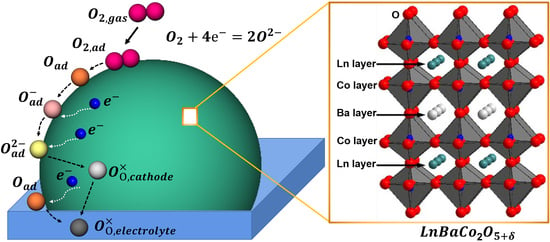
Catalysts | Free Full-Text | Progress in Developing LnBaCo2O5+δ as an Oxygen Reduction Catalyst for Solid Oxide Fuel Cells
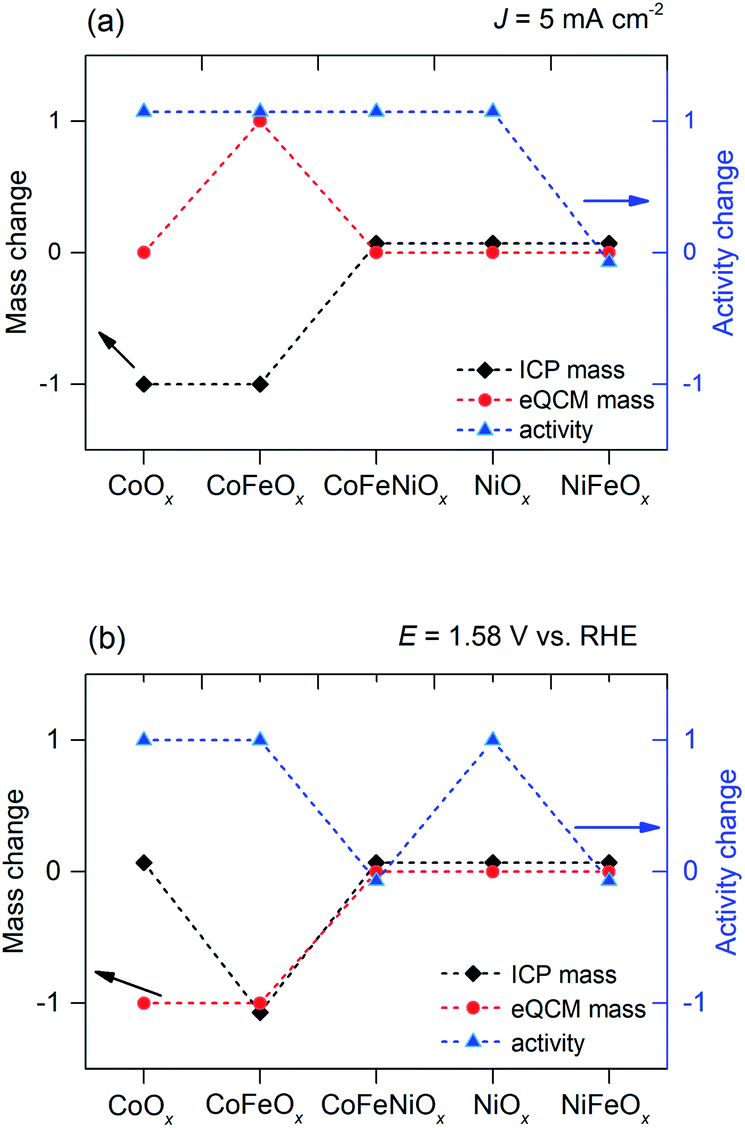
Stability profiles of transition metal oxides in the oxygen evolution reaction in alkaline medium - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/C9TA10308B

Catalysts | Free Full-Text | Synthesis and Characterisation of Cobalt Ferrite Coatings for Oxygen Evolution Reaction

Elucidating the Mechanism of Fe Incorporation in In Situ Synthesized Co–Fe Oxygen-Evolving Nanocatalysts | Journal of the American Chemical Society
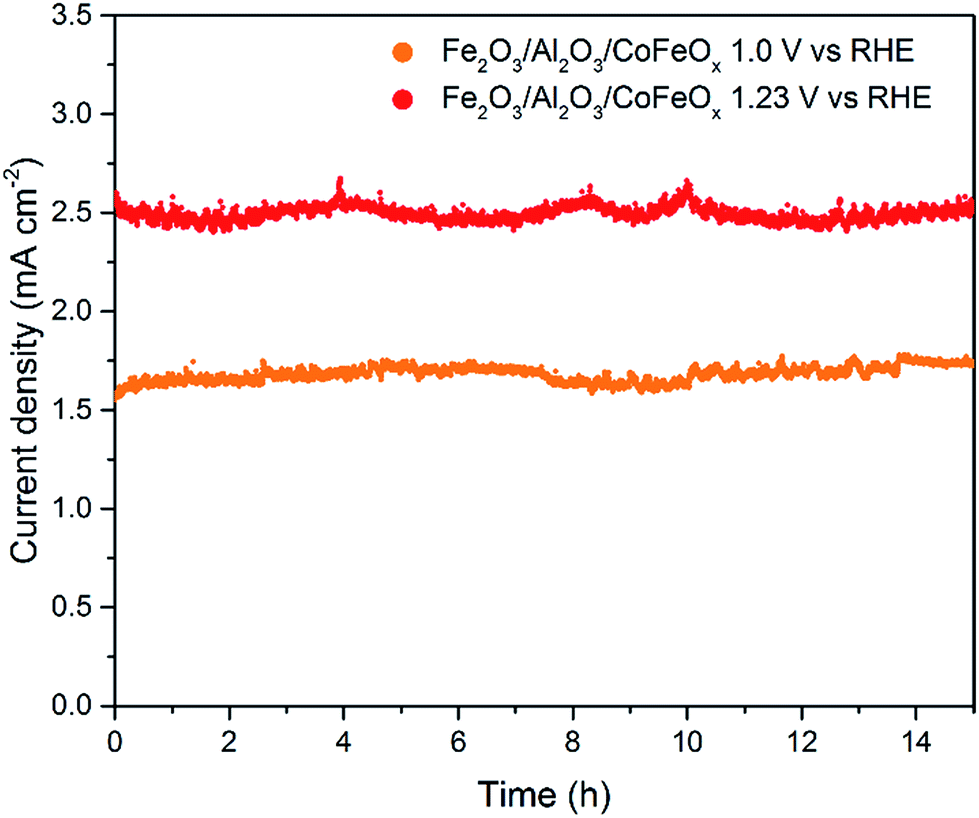
An ultrathin cobalt–iron oxide catalyst for water oxidation on nanostructured hematite photoanodes - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/C8TA12295D

Effects of Structure and Constituent of Prussian Blue Analogs on Their Application in Oxygen Evolution Reaction. - Abstract - Europe PMC

On the Oxidative Valorization of Lignin to High‐Value Chemicals: A Critical Review of Opportunities and Challenges - Abdelaziz - 2022 - ChemSusChem - Wiley Online Library

Comparative Heating Efficiency of Cobalt-, Manganese-, and Nickel-Ferrite Nanoparticles for a Hyperthermia Agent in Biomedicines | ACS Applied Materials & Interfaces

Manipulating the morphology of 3D flower-like CoMn2O4 bimetallic catalyst for enhancing the activation of peroxymonosulfate toward the degradation of selected persistent pharmaceuticals in water - ScienceDirect
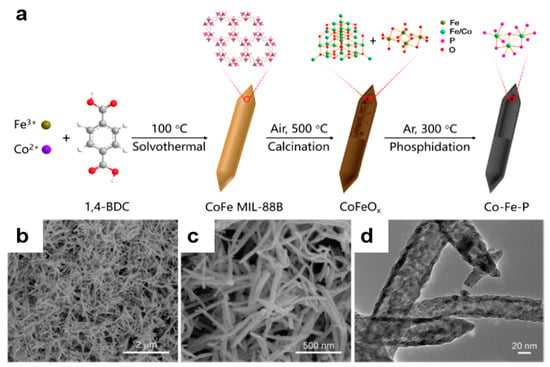
Catalysts | Free Full-Text | Bimetallic Iron–Cobalt Catalysts and Their Applications in Energy-Related Electrochemical Reactions

Catalytic Hydrolysis of Sodium Borohydride for Hydrogen Production Using Magnetic Recyclable CoFe2O4-Modified Transition-Metal Nanoparticles | ACS Applied Nano Materials

Hematite Photoanodes for Water Oxidation: Electronic Transitions, Carrier Dynamics, and Surface Energetics - Li - 2021 - Angewandte Chemie International Edition - Wiley Online Library

A Polymer-Derived Co(Fe)Ox Oxygen Evolution Catalyst Benefiting from the Oxidative Dehydrogenative Coupling of Cobalt Porphyrins | ACS Catalysis

Dynamic Role of Cluster Cocatalysts on Molecular Photoanodes for Water Oxidation | Journal of the American Chemical Society






